May Product Updates
Heads up to our Voicebrook customers! We have a new product release this May to support the College of American Pathologists' recently revised Cancer...
1 min read
 Melanie Shedd
Thursday March 15, 2018
Melanie Shedd
Thursday March 15, 2018

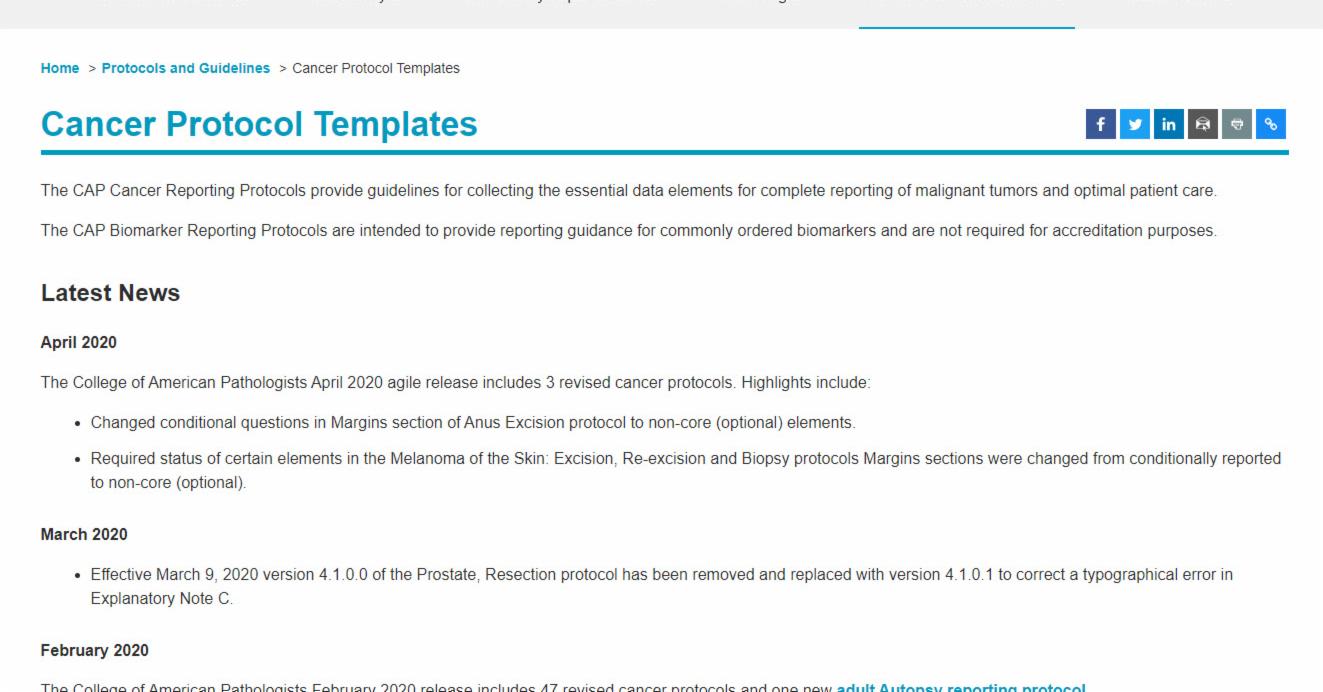
The College of American Pathologists (CAP) is continuously improving its protocols, and it can be a challenge for Pathology departments to maintain their electronic versions and keep pace with the changes. On its website, CAP offers resources and links to partners who can help accredited laboratories stay up to date with the latest versions of the checklists
 On January 30, 2018, CAP released a new version of the Breast Invasive and Breast DCIS Cancer Protocols on their website. The updates for the Breast checklists affect compliance and are required to be in place by September 30th, 2018.
On January 30, 2018, CAP released a new version of the Breast Invasive and Breast DCIS Cancer Protocols on their website. The updates for the Breast checklists affect compliance and are required to be in place by September 30th, 2018.
CAP has released the electronic version of the two Breast checklists, a new optional “DNA Mismatch Repair Biomarker Testing for Checkpoint Inhibitor Immunotherapy” checklist, and made updates across 54 other checklists.
Note from CAP: “The Prostate Needle Biopsy templates are undergoing extensive revisions and are not included in this release. They will be made available at a later time.”
Voicebrook’s March release will include all the CAP changes released for the electronic Cancer Checklists. If you already have SRE, your project manager will get in touch within the next two weeks to schedule the download of the new content.
If you don't have SRE and are interested in how it could simplify your CAP eCC reporting, let us know!
Heads up to our Voicebrook customers! We have a new product release this May to support the College of American Pathologists' recently revised Cancer...

The College of American Pathologists Cancer Protocols have become the standard in cancer reporting. According to their website, “For more than 20...

Healthcare organizations around the world have discovered the benefits of "big data" thanks to the enormous amounts of data available from electronic...